
1a, middle) by incubating equimolar concentrations (5 µM) of Connectase and the fluorescent peptide substrate for 1 min. The formation of a fluorophore-Connectase conjugate (N-Cnt see Fig. 2 C-terminally tagged proteins require a different procedure and are not studied here). Using these components, we established a standard protocol to visualize proteins with N-terminal CnTags (Fig. These could be obtained by routine cloning and purification procedures because the start-methionine preceding the CnTag (PGA.) is removed during protein expression by endogenous methionine aminopeptidase 16, 17. Furthermore, we generated target proteins with N-terminal CnTags (PGAFDADPLVVEI) and short linker sequences (5 amino acids, e.g., AAAGA (see Supplementary Information)). We designed a peptide substrate, with the Cy5.5 fluorophore at the N-terminus, followed by 20 residues based on the Methanosarcina mazei MtrA sequence (Cy5.5-RELAS KDPGAFDADPLVVEI). 
coli cell extract to simulate sample impurities. Sensitive and reliable target protein detectionįor our experiments, we used Connectase from Methanosarcina mazei and performed all reactions at room temperature, neutral pH (7.0–7.5), moderate salt concentrations (150 mM NaCl, 50 mM KCl), and in presence of E. Consequently, an alternative CnTag-substrate can replace the original CnTag-fragment in the reverse reaction, resulting in the formation of a new fusion product (Fig. This reaction is reversible, meaning that the substrate is constantly cleaved and re-ligated. In other words, the N-terminal substrate fragment ending in KD is fused to the enzyme (in the following N-Cnt), while the C-terminal fragment starting with PGA plus another 10 amino acids (in the following CnTag) is cleaved off. Upon binding, Connectase cleaves the amide bond between aspartate and proline in the KDPGA sequence and forms a new amide bond between its own N-terminal amino group and said aspartate. We found that Connectase acts on this recognition sequence, even when it is fused to other proteins or molecules. 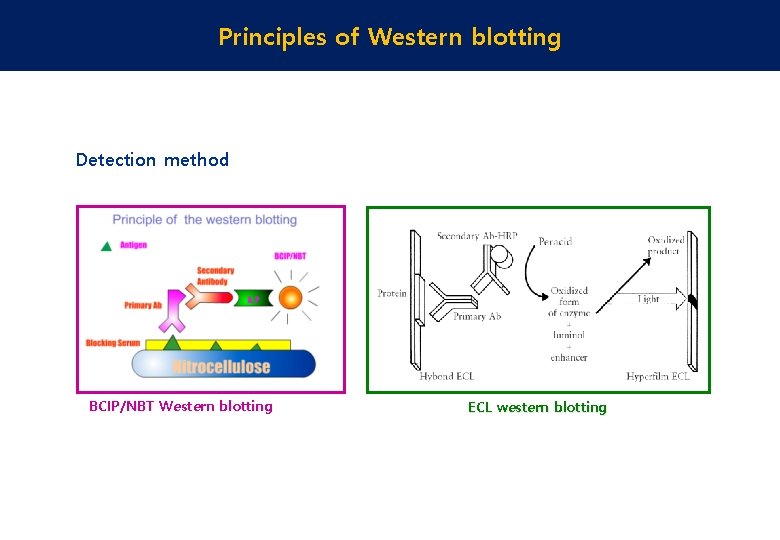
This sequence consists of a highly conserved KDPGA motif and 10 residues C-terminal of that motif, which vary across different species. Here, it recognizes a sequence in its interaction partner Methyltransferase A (MtrA), which links the catalytic and the transmembrane domain. This enzyme, Connectase (Cnt), is found in methanogenic archaea. Recently, we identified a protein ligase with very different characteristics 16. Remarkably, however, no protein ligase-based system has yet been established, primarily because the few known ligase enzymes have a low substrate specificity and suffer from side reactions 15. Alternatively, His 6- 13 or His 12-tags 14 can be detected directly in polyacrylamide gels by fluorophore-conjugated chelator probes. For example, a C-terminal 11 amino acid fragment, the HiBiT tag, can be used to reconstitute a split luciferase in order to visualize the tagged protein on a blotting membrane 12. Similar small protein tags can also be detected by other means. Popular tags include Streptavidin-, FLAG-, V5-, cMyc-, His 6-, HA-, or E-tags, and vary between 6–14 amino acids in length, plus additional linker sequences. As these antibodies are often better characterized and more reliable compared to POI-specific antibodies 8, many researchers prefer this method to study exogenous recombinant proteins, and an increasing number of scientists use gene-editing tools to insert tags in endogenous proteins 9, 10, 11. In such cases, a small peptide tag can be introduced, which is detectable with a tag-specific antibody. While commercial POI-specific antibodies are available for many proteins from model organisms and for well-studied targets, no suitable antibodies are on the market for the majority of proteins. In addition, quantitative analyses are complicated 4 and often result in a hyperbolic rather than in the desired linear signal-to-substrate relationship 1, 4, 7. Even with identical samples and standard operating procedures, results from different users may vary significantly 6. This impedes the comparability of different blots, in particular across different labs 3, 4, 5. Overall, the procedure involves multiple steps, which are often adapted depending on the target protein, the chosen antibody combination, or the sample composition 1, 2. After additional binding sites on this membrane have been blocked, target-specific antibodies are used to detect the protein of interest (POI), and these, in turn, are visualized by reporter-conjugated antibodies. Here, proteins within a complex mixture are separated on a polyacrylamide gel and transferred to a membrane. Western blots present one of the most widely used methods in molecular research.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
